Rapid deployment aortic valves deliver superior hemodynamic performance in vitro
Ai L et al. Innovations. 2017; 12: 338–45. DOI: 10.1097/imi.0000000000000407
Aim
To develop a clinically relevant aortic root model, and then compare the haemodynamic function of the EDWARDS INTUITY valve (a rapid deployment valve) with the Carpentier- Edwards PERIMOUNT Magna Ease valve (a conventional bioprosthetic valve), in vitro.
Background Information
Many trials have shown the short-term safety and efficacy of sutureless rapid deployment aortic valves, but direct comparisons of the haemodynamic performance of these new valves with conventional bioprosthetic valves are lacking.
Methods
Model development
Multi-slice computed tomography reconstructions in patients with aortic stenosis were used to generate a 3D printed model of an aortic root.
Effective diameter of the aortic annulus was 21 mm, effective area was 3.38 cm2 and LVOT area was 3.01 cm2.
Valve Deployment
For measurement of haemodynamic parameters (EOA, TPG and LVOT geometry), Magna Ease valves were sutured to the model’s annulus, with and without the use of pledgets, and EDWARDS INTUITY valves were secured by balloon inflation. EDWARDS INTUITY
valves were further tested with frames expanded
to 22, 23, 24 and 25 mm.
Figure 1: EDWARDS INTUITY and Magna Ease valves in the aortic root model and cadaver heart
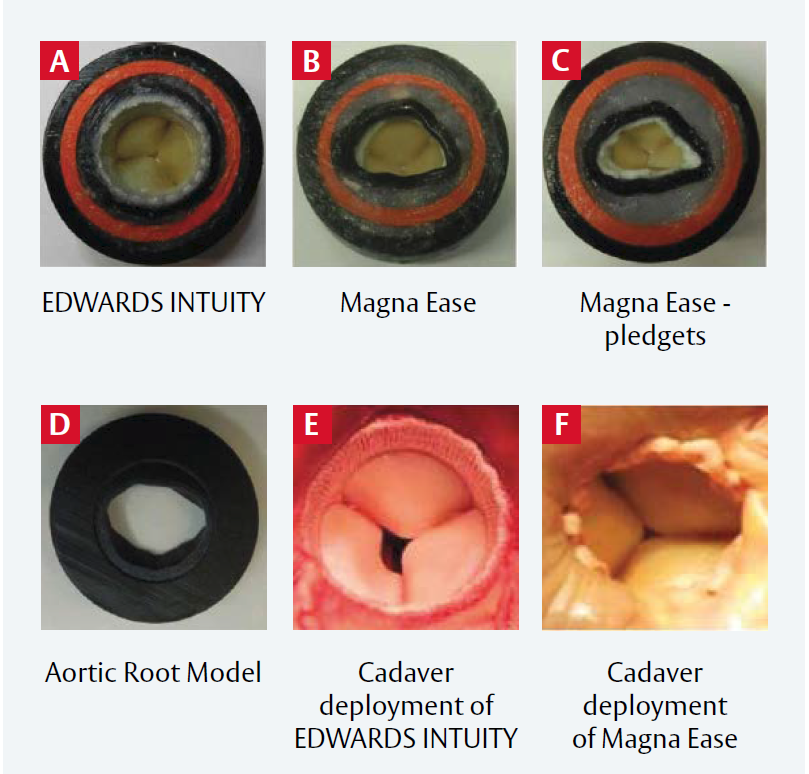
A: EDWARDS INTUITY valve in the model;
B: Magna Ease valve in the model without pledgets;
C: Magna Ease valve in the model, with pledgets;
D: Outflow view of the aortic root model; E: EDWARDS INTUITY valve in the cadaver heart;
F: Magna Ease valve without pledgets in the cadaver heart.
Reproduced from Ai L et al, 2017. © 2017 Wolters Kluwer Health, Inc. Reproduced with permission LVOT in the rubber gasket aortic root model and in the cadaver heart.
Results
Hemodynamic Performance
The EDWARDS INTUITY valve had both a greater EOA and a lower TPG than the Magna Ease valve, irrespective of pledget use (Figure 1). EOA
and TPG were also significantly different between Magna Ease valves with and without pledgets (all p<0.002 or 0.001).
LVOT Performance
Expanding the EDWARDS INTUITY valve frame diameter from 22 to 25 mm increased the average LVOT area index stepwise from 1.07 to 1.34: this minimally affected EOA or TPG (Table 1).
EDWARDS INTUITY valves resulted in more circular LVOTs than Magna Ease valves, although increasing ellipticity did not impact EOA or TPG.
Average maximum flow velocity was lower across the EDWARDS INTUITY valve than the Magna Ease valve without pledgets (p<0.003), as were maximum turbulent shear stress (p<0.02) and turbulent kinetic
energy (p<0.002).
| EDWARDS INTUITY valve (n=4) | Magna Ease valve (n=4) | Magna Ease valve with pledgets (n=4) | |
| EOA, cm2 | 1.85 ± 0.06 | 1.56 ± 0.01 | 1.21 ± 0.08 |
| Mean gradient, mmHg | 16.8 ± 1.3 | 23.4 ± 0.51 | 1.21 ± 0.08 |
EOA: effective orifice area
Table 1: TPG and EOA of models with an EDWARDS INTUITY valve or a Magna Ease valve (with or without pledgets)
Limitations
- Such a model, isolated from the left ventricle, cannot replicate all physiological mechanical properties.
- Pathological changes and normal physiological variations can affect valve performance in vivo: these were not modelled here.
- The use of saline, rather than blood or a substitute, may have affected simulated valve performance.
Conclusions
- Haemodynamic performance was superior for the EDWARDS INTUITY valve than the Magna Ease valve with increased EOA and reduced mean gradient.
- Pledgets worsened Magna Ease valve performance.
- Maximum velocity, turbulent shear stress and kinetic energy were all lower across the EDWARDS INTUITY valve than the Magna Ease valve.
- Haemodynamic advantages may account for the superior clinical performance of rapid deployment valves over conventional surgical valves
Important safety information:
Use of the EDWARDS INTUITY Elite valve system may be associated with new or worsened conduction disturbances,
which may require a permanent cardiac pacemaker implant (PPI). The rate of PPI for the EDWARDS INTUITY Elite
valve is within the range reported in the literature for various rapid deployment valves, but higher than that reported
for surgical aortic valves. Physicians should assess the benefits and risks of the EDWARDS INTUITY Elite valve prior to
implantation. See instructions for use for additional information.
For professional use. For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
Edwards devices placed on the European market meeting the essential requirements referred to in Article 3 of the Medical Device Directive 93/42/EEC bear the CE marking of conformity
Edwards, Edwards Lifesciences, the stylized E logo, Carpentier-Edwards, Carpentier-Edwards PERIMOUNT, EDWARDS INTUITY, EDWARDS INTUITY Elite, Magna, Magna Ease, PERI, PERIMOUNT and PERIMOUNT Magna are trademarks are trademarks or service marks of Edwards Lifesciences Corporation or its affiliates. All other trademarks are the property of their respective owners.
© 2021 Edwards Lifesciences Corporation. All rights reserved. PP–EU-2350 v1.0
Edwards Lifesciences • Route de l’Etraz 70, 1260 Nyon, Switzerland • edwards.com





 ~5m
~5m