A mechanistic investigation of the EDWARDS INTUITY Elite valve’s hemodynamic performance
Keywords: aortic stenosis; aortic valve replacement; transvalvular pressure gradients; valve haemodynamics
Rapid deployment valves requiring minimal sutures are an alternative to conventional surgical valves. In addition to facilitating minimally invasive surgery, rapid deployment valves are associated with improved haemodynamic outcomes compared with conventional surgical valves. The mechanism behind these haemodynamic outcomes is unknown.
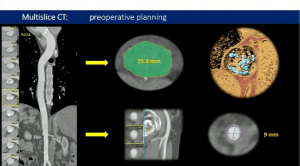
Sadri et al. present the first in-vitro study that compares a conventional valve with a rapid deployment valve in the same surrounding physiology. PERIMOUNT Magna Ease and EDWARDS INTUITY Elite valves were sequentially implanted in a donor heart, and μ-computerised tomography used to develop 3D-printed flow models for each valve. Sadri et al. explain how the EDWARDS INTUITY Elite, a rapid deployment valve, may increase effective orifice area and lower transvalvular pressure gradients, peak velocities and patient─prosthesis mismatch.
Click on the link below to see how two different valves perform in the same heart.
Click here to access the full publication
Important safety information:
Use of the EDWARDS INTUITY Elite valve system may be associated with new or worsened conduction disturbances, which may require a permanent cardiac pacemaker implant (PPI). The rate of PPI for the EDWARDS INTUITY Elite valve is within the range reported in the literature for various rapid deployment valves, but higher than that reported for surgical aortic valves. Physicians should assess the benefits and risks of the EDWARDS INTUITY Elite valve prior to implantation. See instructions for use for additional information.
For professional use. For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
Edwards devices placed on the European market meeting the essential requirements referred to in Article 3 of the Medical Device Directive 93/42/EEC bear the CE marking of conformity.
Edwards, Edwards Lifesciences, the stylized E logo, Carpentier-Edwards, EDWARDS INTUITY, EDWARDS INTUITY Elite, Magna, Magna Ease, PERI, PERIMOUNT, and PERIMOUNT Magna Ease are trademarks or service marks of Edwards Lifesciences Corporation or its affiliates. All other trademarks are the property of their respective owners.
© 2020 Edwards Lifesciences Corporation. All rights reserved.
Edwards Lifesciences • Route de l’Etraz 70, 1260 Nyon, Switzerland • edwards.com





 ~5m
~5m