December 8, 2022
 ~5m
~5mMeuris B, Senage T, Borger M, Siepe […]
INDURE registry: Clinical and quality of life outcomes of surgical aortic valve replacement in 421 patients under 60 years of age
Meuris B, Senage T, Borger M, Siepe M, Stefano P, Laufer G, Langanay T and De Paulis R, on behalf of the INDURE investigators
Presented at 36th European Association for Cardio-Thoracic Surgery Annual Meeting, 2022.
Key points:
- One-year clinical outcomes of patients aged ≤60 years within the INDURE registry were excellent, with few cardiovascular events and satisfactory valve performance
- Patients achieved a general population standard quality of life (QoL) within 3–6 months post surgery
Background information
- The treatment of comparatively young patients with implantation of surgical aortic bioprosthetic valves has raised questions about valve durability and longetivity.1
- The INSPIRIS RESILIA Durability Registry (INDURE) was designed to assess the performance of the INSPIRIS RESILIA bioprosthetic valve in patients aged 60 years or younger.1
Aims
- To evaluate the clinical and QoL outcomes of the INSPIRIS RESILIA valve in patients aged ≤60 years 1 year after surgical aortic valve replacement (SAVR).
Type of study
- A prospective, open-label, multicentre registry study.
Endpoints
Endpoints included time-related valve safety according to VARC-2 guidelines and haemodynamic performance at 1 year and changes in QoL from baseline to 1 year.
- Follow-up included Clinical Events Committee adjudication and CoreLab echocardiography adjudication at 1, 3 and 5 years.
Methods
- The study included patients aged 18–60 years undergoing SAVR.
- Patients were followed-up with echocardiography after 1 year.
- QoL was assessed using the Kansas City Cardiomyopathy Questionnaire (KCCQ) and the Short Form 12 item (version 2) Health Survey (SF12v2).
Results
Patients
- The 1-year analysis included 421 patients from 21 centres across Europe and Canada, with a mean age of 53.5 ± 6.9 years, of whom 23.5% were female. Mean body surface area was 2.00 ± 0.23 m2. Mean EuroSCORE II was 1.5 ± 1.6% and 27.2% of patients were in NYHA class III/IV.
- The mean follow-up was 7 ± 2.3 months.
- Despite being of similar age to males, female patients had reduced body surface area (p<0.001), increased EuroSCORE II (p<0.001), as well as increased incidence of NYHA class III or IV (p<0.001) and chronic obstructive pulmonary disease (p=0.005).
Clinical outcomes
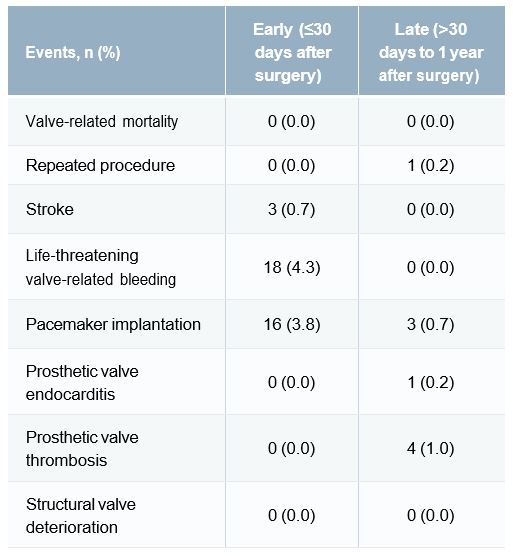
- Clinical outcomes were excellent (Table 1), with a hospital mortality that was better than predicted (0.7% vs 1.5%).
- Haemodynamic performance was satisfactory and stable throughout follow-up (mean pressure gradient at discharge was 7 ± 4.3 mmHg, and 12.6 ± 5.3 mmHg at one year).
- No cases of structural valve deterioration (SVD) were recorded.
Table 1. Safety outcomes early and between 31 days and 1 year after surgery
QoL
- Within 3–6 months, QoL was restored to good-to-excellent on the KCCQ scale (score between 75–100) and approximately matching the QoL of the general US population according to the SF12v2 scoring system (approximate score of 50).
- Female patients had generally significantly lower scores than men.
Conclusion
One-year clinical outcomes of patients aged ≤60 years within the INDURE registry were excellent, with stable performance, better-than-predicted hospital mortality, low incidence of cardiovascular events, only four incidences of valve thrombosis (which did not require intervention), and no SVD events. Patients returned to expected QoL within 3–6 months after surgery. Notably, female patients tended to have a higher NYHA functional class at the time of surgery and had consistently lower QoL scores than males. Longer follow-up is underway.
References
- Meuris B, Borger MA, Bourguignon T et al. Durability of bioprosthetic aortic valves in patients under the age of 60 years – rationale and design of the international INDURE registry. J Cardiothorac Surg. 2020; 15: 119.
This document is a summary of the Meuris B et al. presentation and covers key information including aim, type of study, methods, results and conclusions.
The full publication is available at:
Abbreviations:

No clinical data are available that evaluate the long-term impact of RESILIA tissue in patients.
Medical device for professional use. For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
Edwards, Edwards Lifesciences, the stylized E logo, INSPIRIS, INSPIRIS RESILIA, and RESILIA are trademarks or service marks of Edwards Lifesciences Corporation. All other trademarks are the property of their respective owners.
© 2022 Edwards Lifesciences Corporation. All rights reserved. PP–EU-5306 v1.0
Edwards Lifesciences • Route de l’Etraz 70, 1260 Nyon, Switzerland • edwards.com
Edwards Masters App
Learn anywhere

now fully customized to your needs and interests. Your educational platform
in heart valve surgery.