Patients: At the heart of what you do
Dr. Vinod Thourani
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Michele De Bonis
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Gilles Dreyfus
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Nikolaos Bonaros
Be courageous, be experienced, be ethical … according to the Guidelines!
Mr. Steve Livesey
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Hanneke Takkenberg
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Bart Meuris
Be courageous, be experienced, be ethical … according to the Guidelines!
Dr. Alison Duncan
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Augusto D’Onofrio
Be courageous, be experienced, be ethical … according to the Guidelines
Prof. Gino Gerosa
Be courageous, be experienced, be ethical … according to the Guidelines!
Prof. Michael Borger
Be courageous, be experienced, be ethical … according to the Guidelines!
The highly anticipated 2021 European Society of Cardiology (ESC) and European Association for Cardio-Thoracic Surgery (EACTS) guidelines for the management of valvular heart disease have finally landed.
We are excited to bring you a summary of the main updates, plus the new materials coming soon to support you and your teams in understanding and implementing the changes.
Key points include:

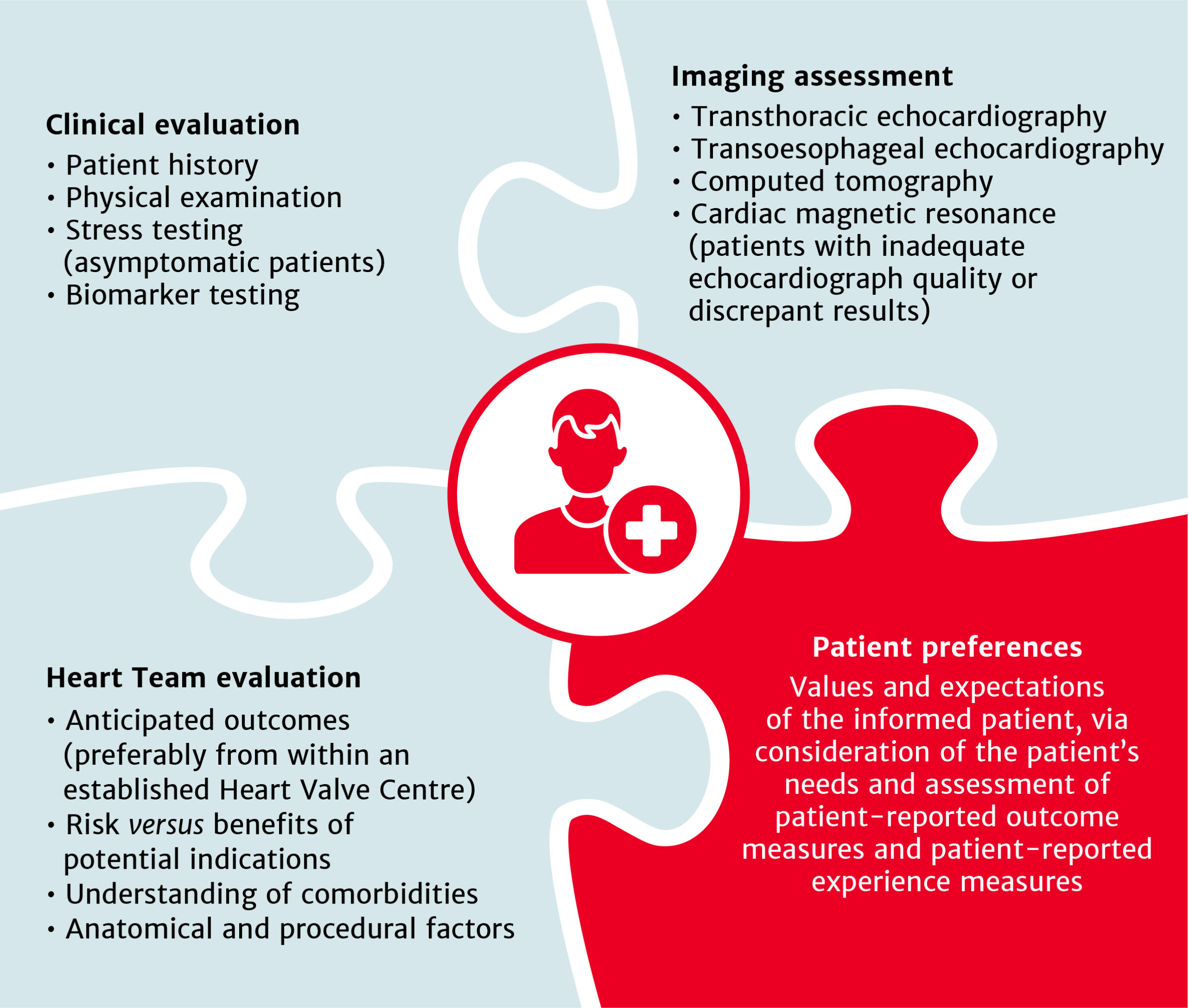
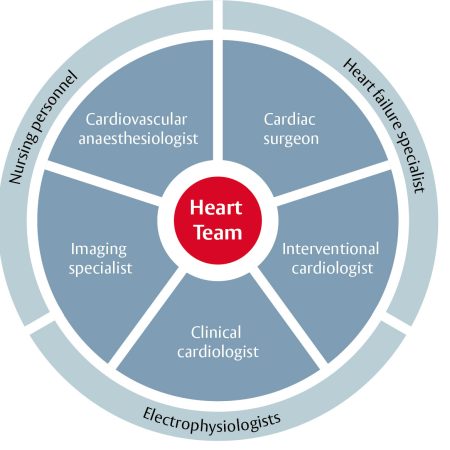
Treatment recommendations for all patients with severe aortic stenosis to be made by the Heart Team

Intervention options for patients with asymptomatic aortic stenosis

Bioprosthetic valves now recommended for those with life expectancy shorter than valve durability

Intervention recommended for patients with secondary mitral regurgitation, who remain symptomatic despite guideline-directed medical therapy
Use your experience of severe aortic stenosis to ensure the best patient outcomes
The choice between surgical aortic valve replacement (SAVR) and transcatheter aortic valve implantation (TAVI) must be made following evaluation of all factors – clinical, anatomical and procedural – weighing the risks and benefits of each approach for all patients with severe aortic stenosis, and after discussion with the patient.

Treatment options have broadened for patients with asymptomatic aortic stenosis, including those with systolic left ventricular (LV) dysfunction (LV ejection fraction <55%) without another cause.
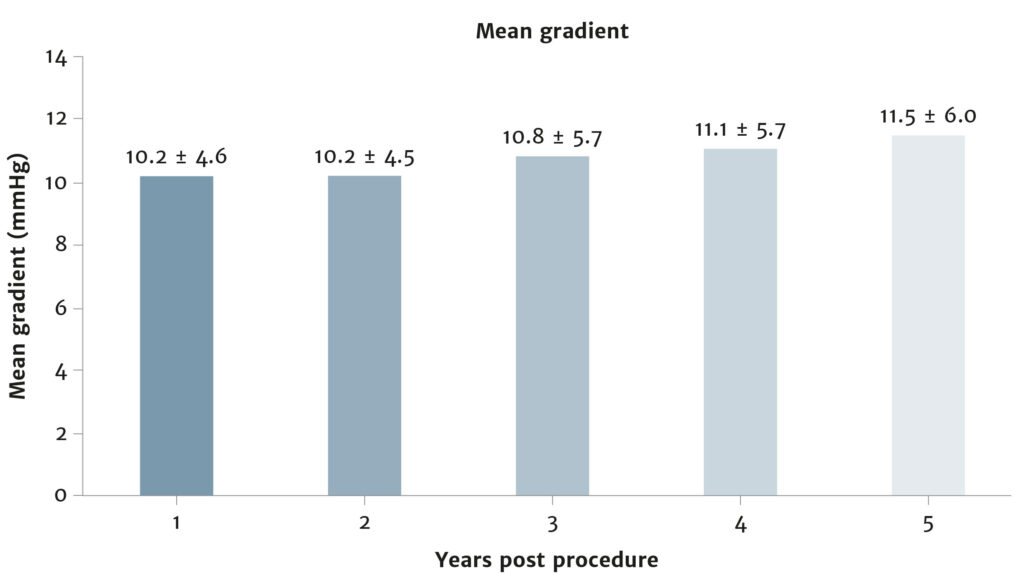
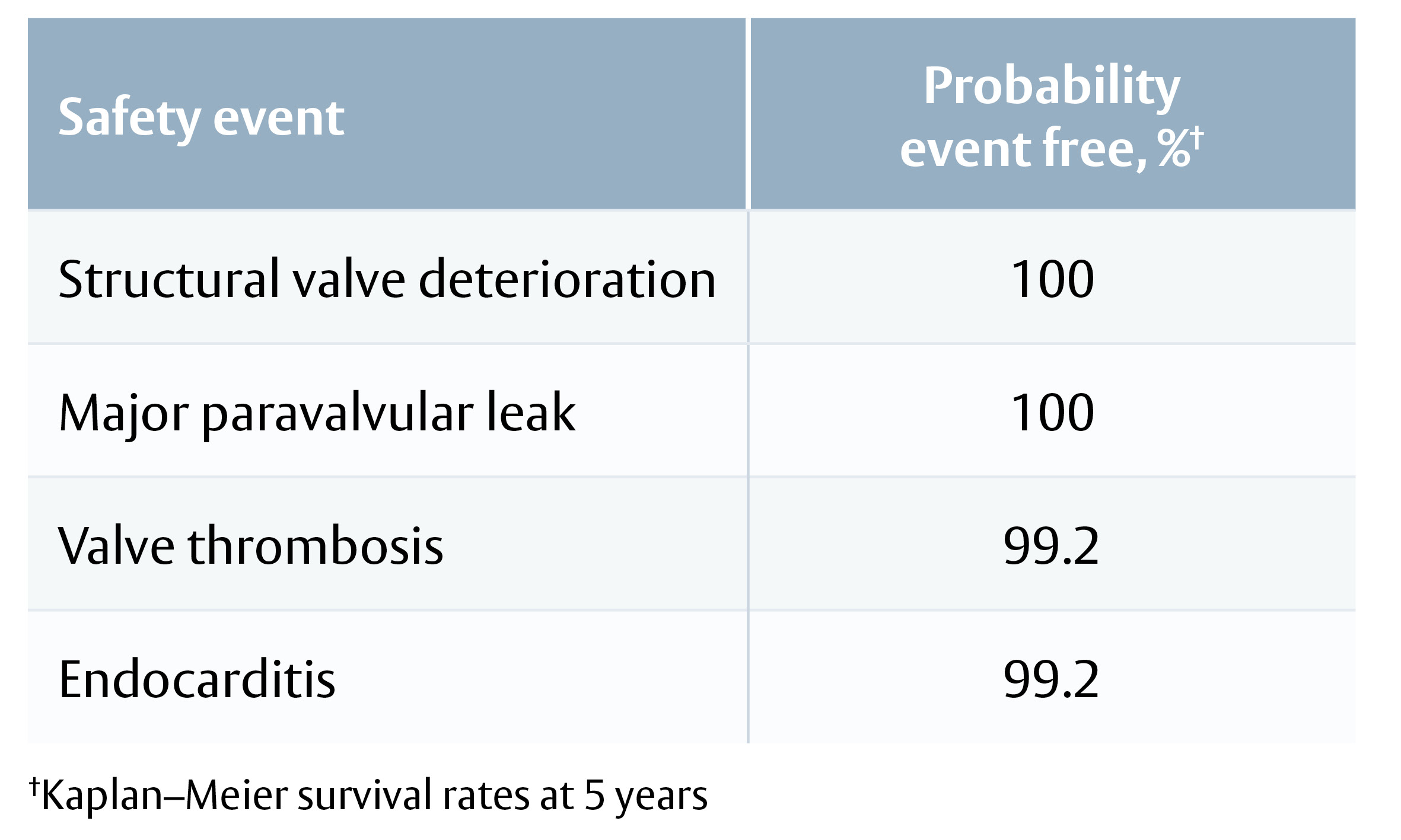
Enable more of your patients to live the active lifestyle they desire
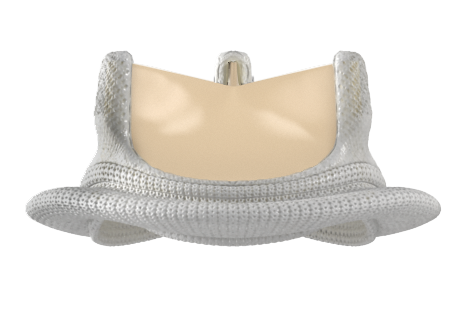
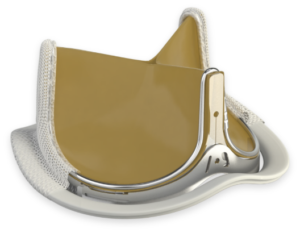
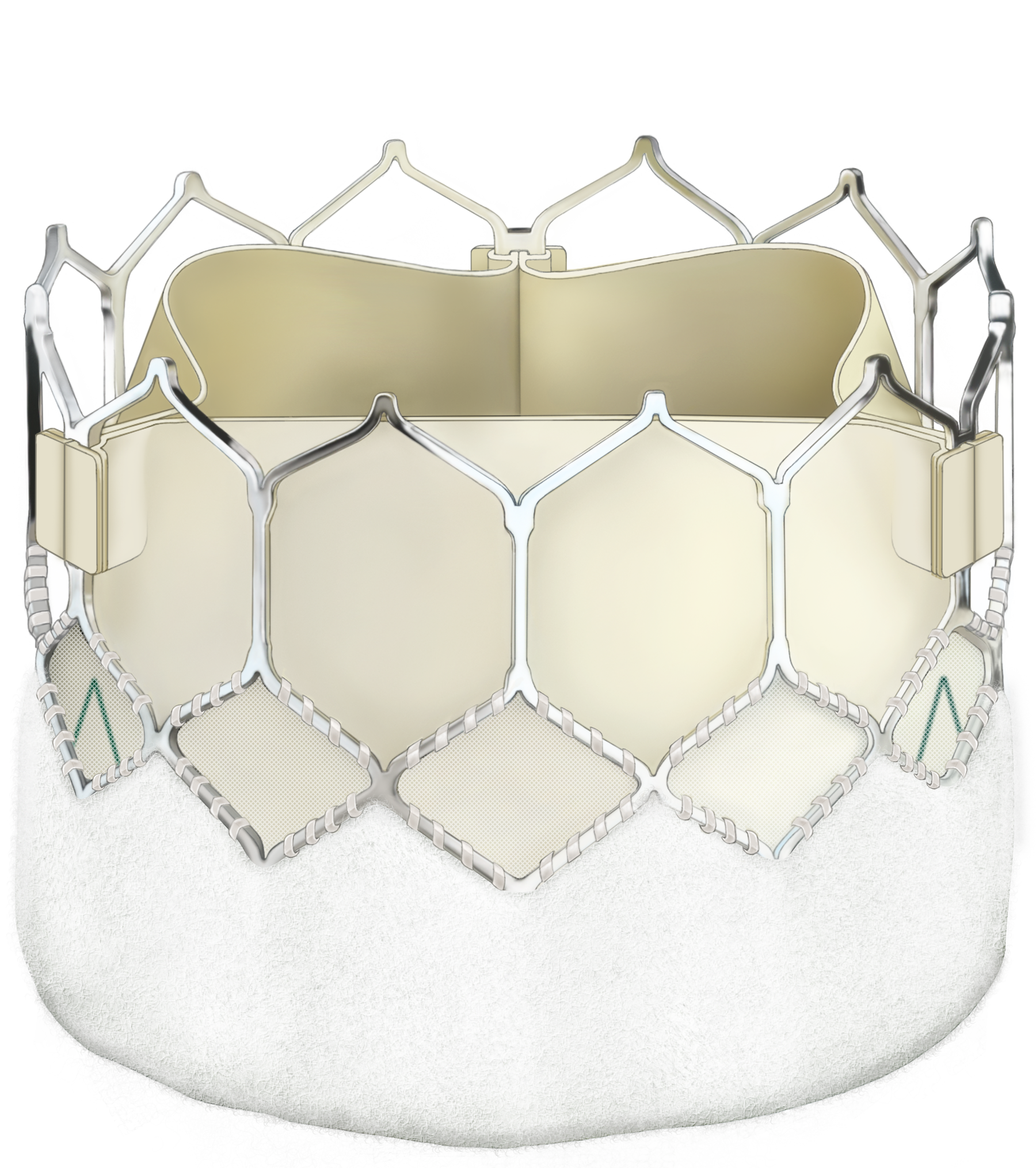
Bioprosthetic valves now have a Class I recommendation for patients with life expectancy lower than the durability of the valve. Now, more of your patients can live a life free from anticoagulation. In addition, bioprosthetic valves may be considered for patients already on long-term novel oral anticoagulants.
Ensure you meet your patients’ high expectations by offering them the best valve technologies.
Be courageous, be experienced, be ethical… according to the guidelines!
Join us from the comfort of your office for a virtual symposium discussing the changes and what they mean for your practice. Eleven speakers from across Europe will cover the implications for the aortic, mitral and tricuspid valves. Click here for the full programme.
Tuesday 21 September 2021
3.00–7.45 pm CET
REGISTER NOW
Coming soon from Edwards
Edwards, Edwards Lifesciences, and the stylized E logo are trademarks or service marks of Edwards Lifesciences Corporation or its affiliates. All other trademarks are the property of their respective owners.
© 2021 Edwards Lifesciences Corporation. All rights
reserved. NP–EU-0633 v1.0
Edwards Lifesciences • Route de l’Etraz 70, 1260 Nyon, Switzerland • edwards.com
Prof. Stephan Ensminger
MVT Aortic 2021