Outcomes 2 Years After Transcatheter Aortic Valve Replacement in Patients at Low Surgical Risk
Leon, Martin B., et al., Journal of the American College of Cardiology (2021) DOI:10.1016/j.jacc.2020.12.052
Aim
Report 2-year findings from PARTNER 3, emphasizing the clinical outcomes from 1 to 2 years and using new standardized definitions of hemodynamic valve deterioration and bioprosthetic valve failure
Methods
- 1000 patients randomly assigned (1:1) to transfemoral TAVR with SAPIEN 3 vs. SAVR
- Mean STS score: 1.9%; mean age: 73 years
- Primary endpoint: composite of death from any cause, all stroke or cardiovascular rehospitalization at 1 year
Results
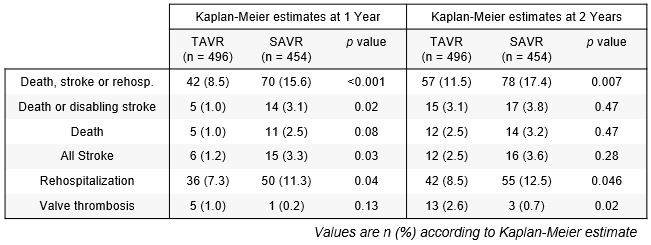
- Overall, complete primary endpoint follow-up through 2 years of 96.5% (TAVR 99%, SAVR 93.8%)
- Functional status, as measured with KCCQ-OS, remained superior for TAVR at 2 years
- Mean gradients were slightly higher after TAVR vs. SAVR (13.6 vs. 11.8 mmHg, p = 0.06; n = 495 and 453 for TAVR and SAVR, respectively) Bioprosthetic valve durability determined using VARC 3 criteria was similar for the 2 groups
Conclusions
- The 2-year follow-up from the PARTNER 3 low-risk trial showed continued superiority of the primary endpoint favoring TAVR versus surgery, but more frequent deaths, strokes, and valve thrombosis events in the TAVR group between 1 and 2 years
- Disease-specific health status at 2 years was better after TAVR than surgery
- Echocardiographic findings through 2 years indicated stable valve hemodynamics and no differences in valve durability parameters
Key talking points
Is there a trend for similar outcomes after TAVR and surgery?
Primary endpoint remained significantly lower with TAVR vs. SAVR, yet the differences in death and stroke have greatly diminished at 2 years. It must be noted that 3 of the 7 deaths occurring between 1- and 2-year follow-up in the TAVR arm were of noncardiac origin (cancer, suicide and sepsis), and 3 of the 6 strokes were disabling. This more granular data points to similar rates of event between year 1 and2 for TAVR and SAVR.
Why was valve thrombosis higher in TAVR patients?
75% of the patients with clinical events committee-adjudicated valve thrombosis were asymptomatic. A computed tomography substudy is running in parallel with the PARTNER 3 trial, increasing awareness on hemodynamic changes and inflating the frequency of valve thrombosis events. The consequences at long term remain uncertain.
Is TAVR the treatment of choice in low-risk patients?
Results of the PARTNER 3 trial are not applicable to all patients at low surgical risk. Exclusion criteria such as bicuspid aortic valves, severe coronary artery disease, concomitant valve disease or vascular disease precluding transfemoral access limit application of these results in these cohorts. Longer follow-up is also needed to assess valve’s durability in younger patients with severe AS.
For professional use. For a listing of indications, contraindications, precautions, warnings, and potential adverse events, please refer to the Instructions for Use (consult eifu.edwards.com where applicable).
Edwards devices placed on the European market meeting the essential requirements referred to in Article 3 of the Medical Device Directive 93/42/EEC bear the CE marking of conformity
Edwards, Edwards Lifesciences, the stylized E logo, PARTNER, PARTNER 3, SAPIEN and SAPIEN 3 are trademarks or service marks of Edwards Lifesciences Corporation or its affiliates. All other trademarks are the property of their respective owners.
© 2021 Edwards Lifesciences Corporation. All rights reserved.
PP–EU-1734 V1.0
Edwards Lifesciences • Route de l’Etraz 70, 1260 Nyon, Switzerland • edwards.com





 ~5m
~5m